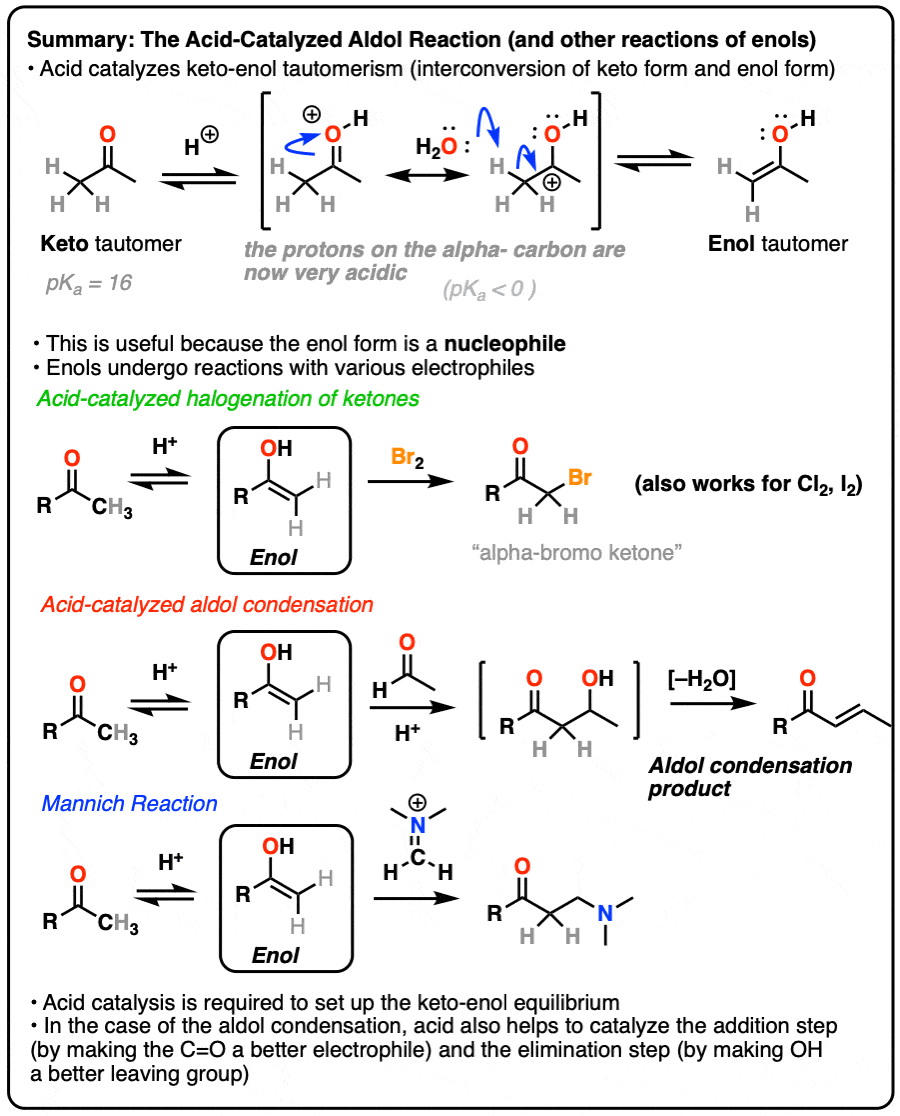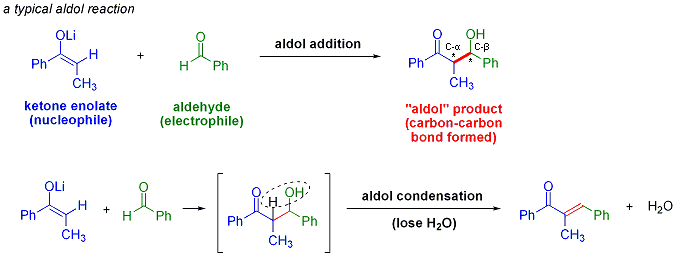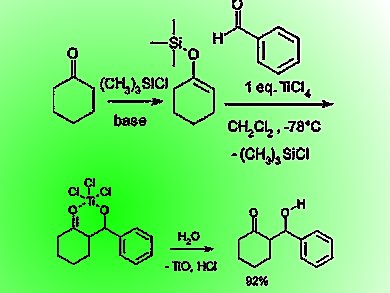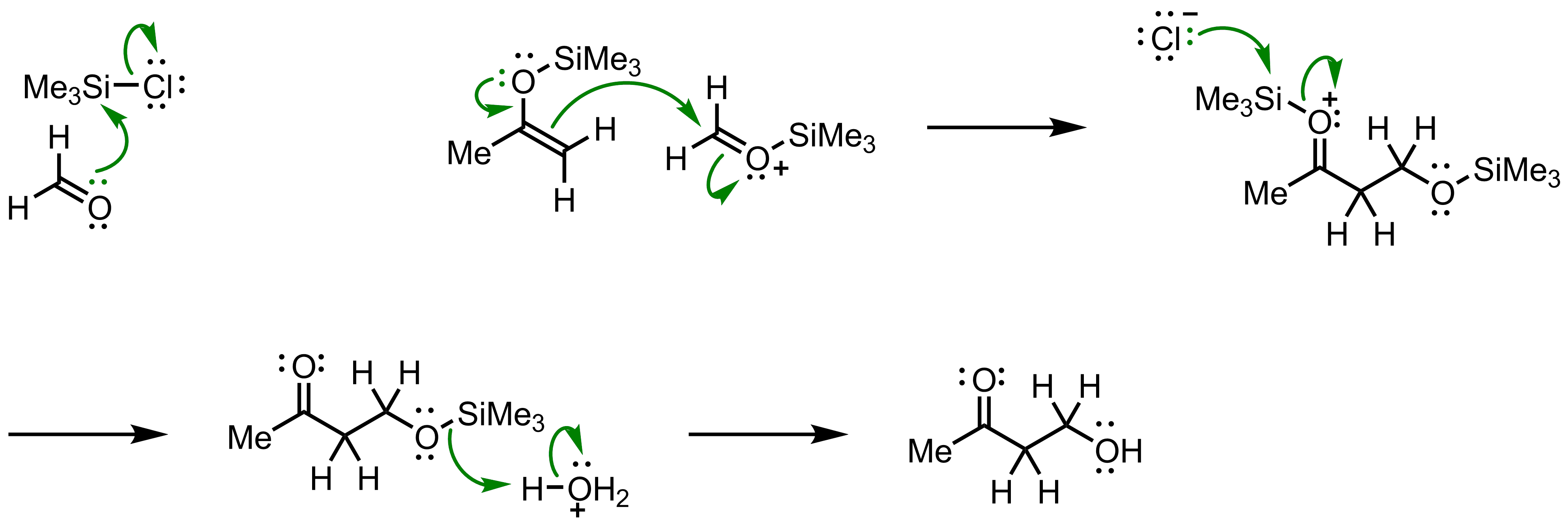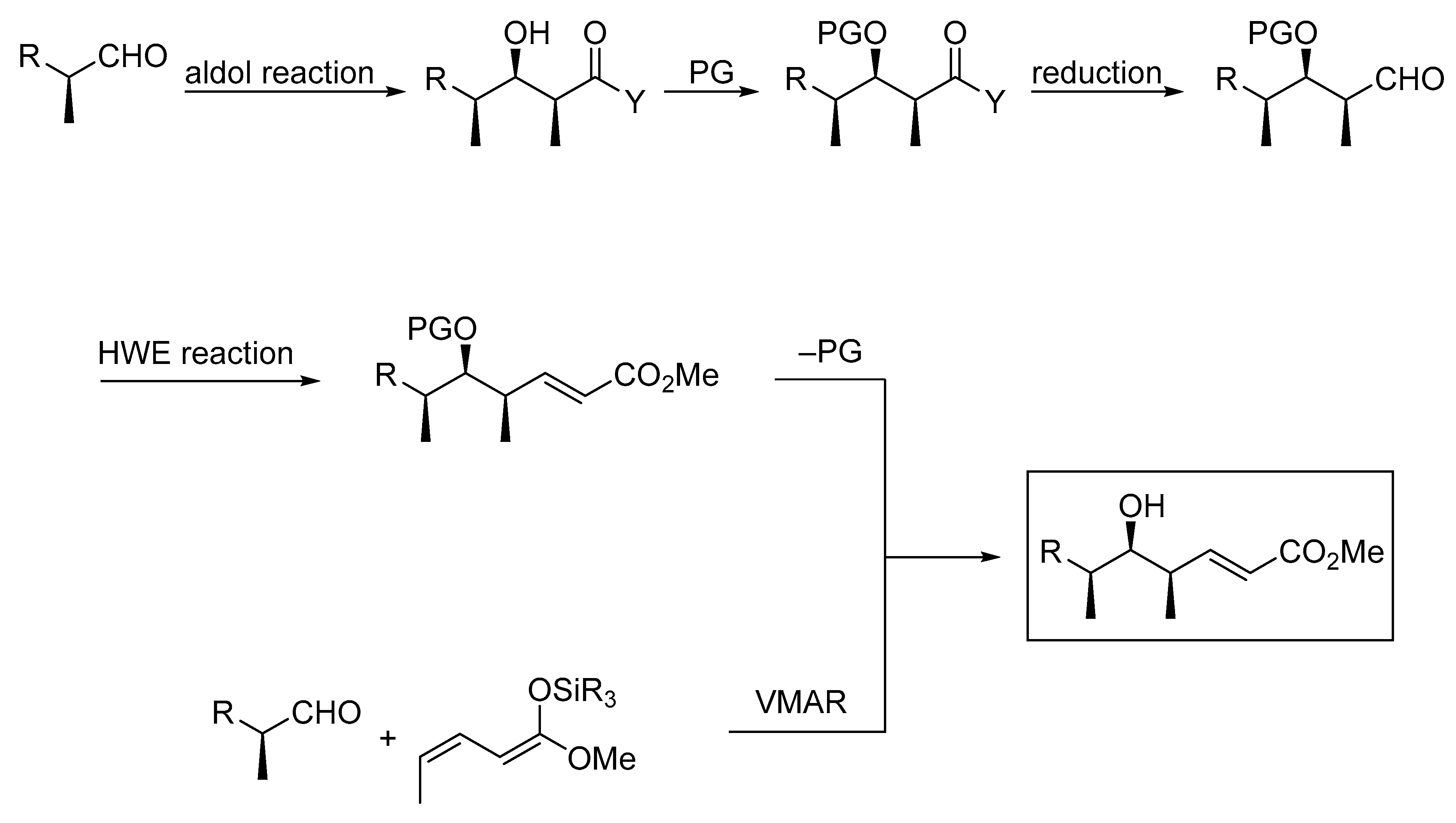
Molecules | Free Full-Text | Very Recent Advances in Vinylogous Mukaiyama Aldol Reactions and Their Applications to Synthesis

Diastereoselectivity in Lewis-acid-catalyzed Mukaiyama aldol reactions: a DFT study. - Abstract - Europe PMC
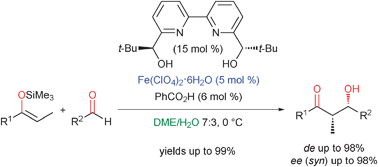
Highly enantioselective Mukaiyama aldol reaction in aqueous conditions using a chiral iron(ii) bipyridine catalyst - Chemical Communications (RSC Publishing)

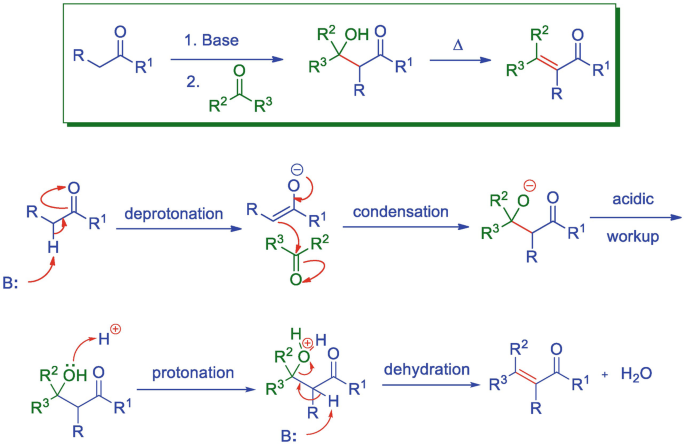
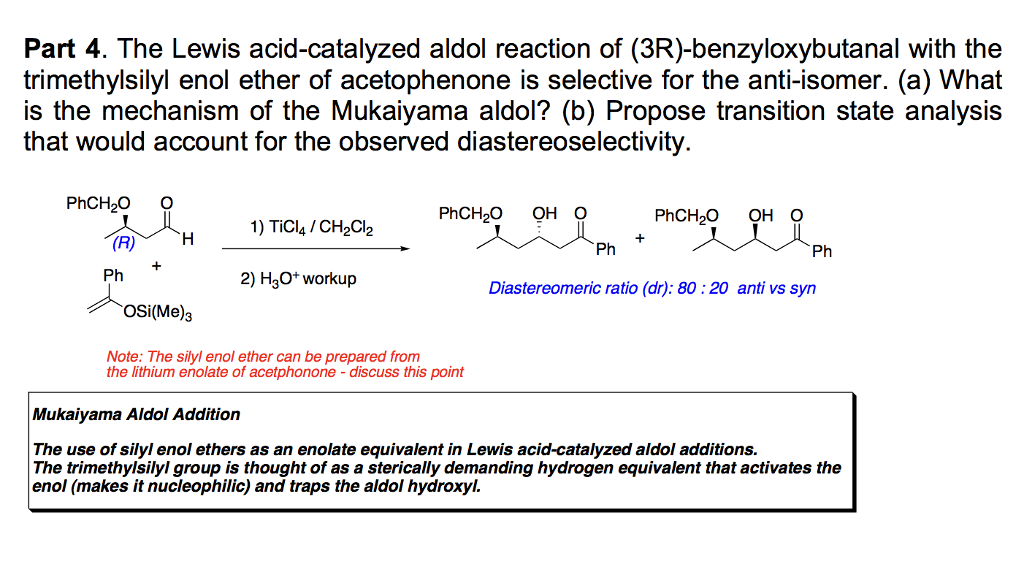
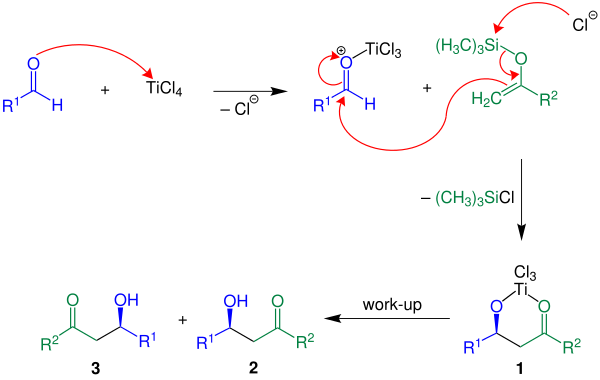
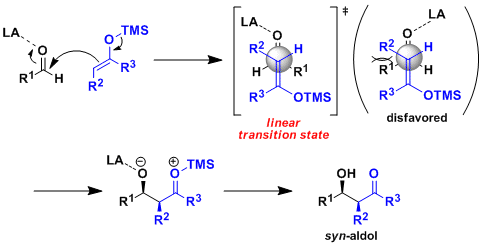
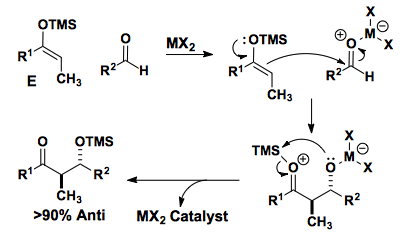
The Mukaiyama Aldol addition is another reaction that provides the functionalization of an aldehyde using a silyl enol ether. Draw the mechanism of the product formation and provide a transition state in

Diastereoselectivity in Lewis-acid-catalyzed Mukaiyama aldol reactions: a DFT study. | Semantic Scholar

Diastereoselectivity in Lewis-Acid-Catalyzed Mukaiyama Aldol Reactions: A DFT Study | Journal of the American Chemical Society